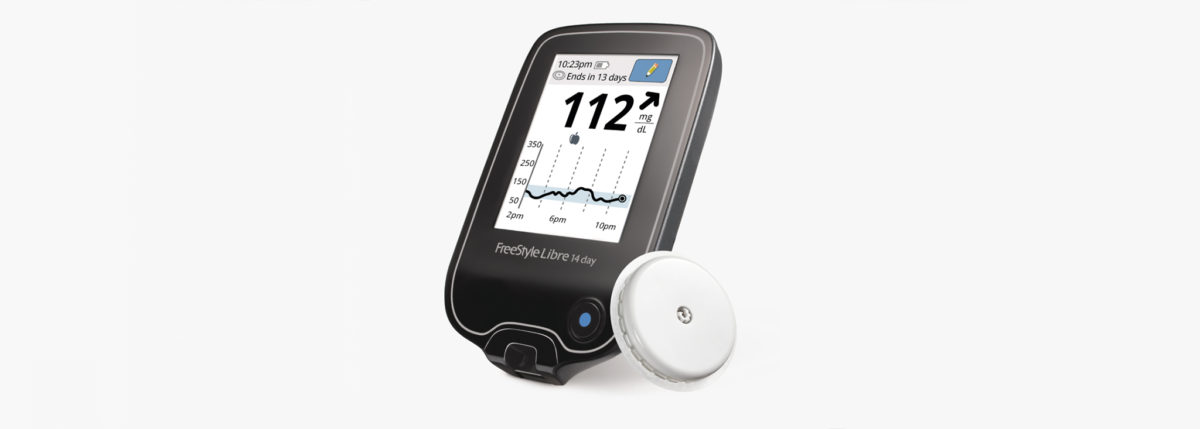
FreeStyle Libre System Available to Medicare Patients
ABBOTT PARK, Ill. – It was just a few months ago that the U.S. Food and Drug Administration approved Abbott Diabetes Care’s FreeStyle Libre Flash Glucose Monitoring System for public use in the U.S., making it the first FDA-approved continuous glucose monitor (CGM) not to require a fingerstick test or manual data entry for calibration. That means that the device is so accurate, people managing type 1 or type 2 diabetes can dose insulin off of it.
Today, Abbott announced that the new, factory-calibrated technology is now available to Medicare patients since its approval by the U.S. Centers for Medicare & Medicaid Services (CMS). It will be the only CGM system available to Medicare patients that doesn’t require calibration.
“At Abbott, we are continuously challenging ourselves to ensure our innovative technology is accessible to the majority of people who need it,” said Jared Watkin, Sr. VP at Diabetes Care. “This revolutionary health technology […] is ultimately going to empower Medicare beneficiaries with diabetes to live better, healthier lives.”
FreeStyle Libre system reads blood glucose levels through a sensor that is worn on the back of the upper arm for up to 10 days, making it the longest lasting personal blood glucose sensor available in the U.S.
Other features include water-resistance and an ability to obtain a reading without the interference of acetaminophen, an active ingredient in many over-the-counter medications, which can inaccurately report dangerously high blood glucose levels.
The FreeStyle Libre also is a system with only two parts: sensor and reader, as opposed to having the the additional transmitter.
Medicare patients can access the FreeStyle Libre system with a prescription through the following suppliers: Edgepark Medical Supplies, Byram Healthcare, Solara Medical Supplies, Edwards Health Care Services, Better Living Now and Mini Pharmacy in the U.S.
Not a Medicare patient? You can purchase the system without insurance coverage via any of the following major U.S. retail pharmacies: CVS, Walgreens, RiteAid, Walmart and Kroger.
George Grunberger, M.D., chairman, Grunberger Diabetes Institute commented, “The accessibility and affordability of this CGM technology is unprecedented, and this is a huge step forward for the diabetes community—both type 1 and type 2.”
From Abbott
INDICATIONS AND IMPORTANT SAFETY INFORMATION
The FreeStyle Libre Flash Glucose Monitoring system is a continuous glucose monitoring (CGM) device indicated for replacing blood glucose testing and detecting trends and tracking patterns aiding in the detection of episodes of hyperglycemia and hypoglycemia, facilitating both acute and long-term therapy adjustments in persons (age 18 and older) with diabetes. The system is intended for single patient use and requires a prescription.
CONTRAINDICATIONS: Remove the sensor before MRI, CT scan, X-ray, or diathermy treatment.
WARNINGS/LIMITATIONS: Do not ignore symptoms that may be due to low or high blood glucose, hypoglycemic unawareness, or dehydration. Check sensor glucose readings with a blood glucose meter when Check Blood Glucose symbol appears, when symptoms do not match system readings, or when readings are suspected to be inaccurate. The FreeStyle Libre system does not have alarms unless the sensor is scanned, and the system contains small parts that may be dangerous if swallowed. The FreeStyle Libre system is not approved for pregnant women, persons on dialysis, or critically-ill population. Sensor placement is not approved for sites other than the back of the arm and standard precautions for transmission of blood borne pathogens should be taken. The built-in blood glucose meter is not for use on dehydrated, hypotensive, in shock, hyperglycemic-hyperosmolar state, with or without ketosis, neonates, critically-ill patients, or for diagnosis or screening of diabetes. Review all product information before use or contact Abbott Toll Free (855-632-8658) or visit www.freestylelibre.us for detailed indications for use and safety information.