COVID-19 + Type 1 Diabetes: New Research Explores Possible Connections
Coverage of the American Diabetes Association (ADA) Scientific Sessions is brought to you by the ADA x BT1 Collab.
Speakers for this session, “COVID-19 and Diabetes,” included: Shuibing Chen, Ph.D., (Weill Cornell Medical College) and co-founder of Oncobeat Inc., W. James Melvin, MD, (University of Michigan) and James Lo, MD, Ph.D., (Weill Cornell Medicine).
In this article, we focus on the research presented by Chen.
Since 2019, scientists and communities have been learning more about the impact of COVID-19 on the human body. Over these past couple of years, there has been ample discussion about how it correlates with autoimmune conditions, like type 1 diabetes.
In a presentation at the American Diabetes Association’s (ADA) 82nd Scientific Sessions, a group of prominent researchers presented their findings on the impact of COVID-19 on people with diabetes and whether contracting it poses a risk of developing diabetes.
Does COVID-19 present a risk of developing type 1 diabetes?
In the first half of this session, Shuibing Chen, Ph.D., presented research conducted by her and her team on “SARS-CoV-2 and Human Islets.”
Chen specializes in understanding Cells that make insulin, a hormone that controls the level of glucose in the blood.beta cells, especially in the context of SARS-CoV-2 (commonly known as COVID-19).
Since COVID-19 is a A disease that affects multiple organs.multi-organ disease, different human “Organoids are tiny, self-organized three-dimensional tissue cultures that are derived from stem cells.” https://hsci.harvard.edu/organoidsorganoid models are used to answer various questions regarding COVID-19, such as:
- Permissiveness: Which organs and tissues can be affected by COVID-19?
- Host response (how your body responds): How do organs and tissues that are affected by COVID-19 respond?
- “Immune-mediated diseases is a group of conditions that result from abnormal activity of the immune cells, overreacting or attacking to the body, displaying an extreme inflammatory response or loss of the ability to recognize and fight against tumor cells.” (FrontiersIn.org)Immune-mediated host damage (how your body attacks itself): COVID-19 infection is only one part of the story. The models that Chen and her team set up analyze how immune cells contribute to host damage.
- “A drug screen (also called a drug test) is the collection and analysis of blood, urine, hair, or saliva to detect the presence of the chemicals and contaminants left behind in the body due to drug use.” (Concentra.com)Drug screening: Chen explained that the advantage of human proponents is that they are a limited resource to derive the types of cells researchers are interested in, which they can use to adapt to a drug screening platform that can look for small markers that can block infection, protect the cells and block the immune-mediated response.
Permissiveness: Multiple organs impacted by COVID-19
The abstract of a featured study in the “permissiveness” section of this presentation illustrated the following:
- COVID-19 impacts the lungs and other organ systems, including the gut, liver, heart and pancreas.
- A “Spike-enabled pseudo-entry virus” infects pancreatic endocrine cells and other cells, neurons,and organoids.
- “Recent clinical studies show a strong association with COVID-19 and diabetes…human pancreatic beta cells and liver organoids are highly permissive (highly impacted by) to SARS-CoV-2 infection.”
Chen explained that high Luciferase is a type of dye used to stain cells to make them glow, essentially. It can also be used as a control. More luciferase activity indicates more cell activity.luciferase activity suggests that the cell can be infected, and there is still discussion in the field as to whether SARS-CoV-2 can be detected in a heart autopsy. (This was the last experiment Chen and her team did before New York City shut down due to the COVID-19 pandemic.)
This study demonstrated that beta cells could be infected “In vivo refers to when research or work is done with or within an entire, living organism. Examples can include studies in animal models or human clinical trials.” (Healthline.com)in vivo, but another study also supported Chen’s results and showed that infection can also be seen “In vitro is used to describe work that’s performed outside of a living organism.” (Healthline.com)in vitro.
After the shutdown and discovering that human islet cells could be infected, Chen and her colleagues, Xuming Tang and Dongxiang Xue, conducted a more thorough study focused on the mechanism behind how COVID-19 impacts pancreatic beta cells. This study checked for other COVID-19 entry factors for human islets. They found that “Nrp1 mediates contacts between the dendritic cells and T cells via homotypic interactions and is essential for the initiation of the primary immune response.” https://www.sciencedirect.com/topics/medicine-and-dentistry/neuropilin-1NRP1, FURIN, and CTSL genes were highly expressed in COVID-19 infected beta cells, leading to lower insulin expression. You can see how that looks here.
When SARS-CoV-2 was intentionally injected (in vitro) in human islet cells as part of this study, multiple clusters of SARS-CoV-2 were detected in a beta population of cells. Some other cell clusters were also infected. A pancreatic autopsy sample of a COVID-19 patient showed some insulin-positive cells were infected with SARS-CoV-2.
Host response: How does the human body respond?
Regarding host response (how the human body responds to COVID-19), the first thing that was checked was the insulin gene expression in beta cells—how much the insulin gene was expressed in the beta cells when a SARS-CoV-2 infection was present. Chen and her team found a decrease in insulin expression in beta cell populations in patients infected with SARS-CoV-2.
The next thing Chen and her team wanted to know was the trajectory of the beta cells. They tried to determine when the expression of insulin decrease occurred in patients infected with SARS-CoV-2. Through this, they also discovered an increase in glucagon.
This graphic summarizes the mechanism in which SARS-CoV-2 causes beta cells to change (transdifferentiate), leading to decreased insulin expression and increased glucagon expression.
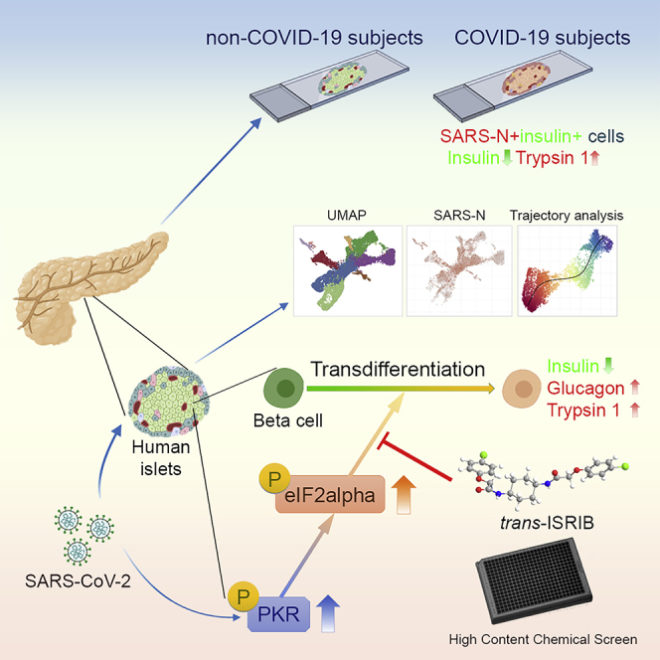
Image Source: Cell Metab. 2021 Aug 3; 33(8): 1577–1591.e7. doi: 10.1016/j.cmet.2021.05.015
Chen explained they could only confirm part of the “The set of observable characteristics of an individual resulting from the interaction of its genotype with the environment.” (Oxford Dictionary)phenotype in the autopsy COVID-19 samples—they failed to establish the presence of glucagon.
While these findings show a connection between beta-cell function and SARS-CoV-2, there are still no definitive answers as to whether COVID-19 directly causes diabetes. The latest updates on COVID-19’s impact on the development of diabetes can be found and should be referenced here.
Editor’s note: The remaining information featured in Chen’s part of the presentation cannot be posted because it is currently unpublished. As/if it becomes available, Beyond Type 1 will publish a follow-up article. Access the entire research paper on “Human organoid models to study SARS-CoV-2 infection” here.





