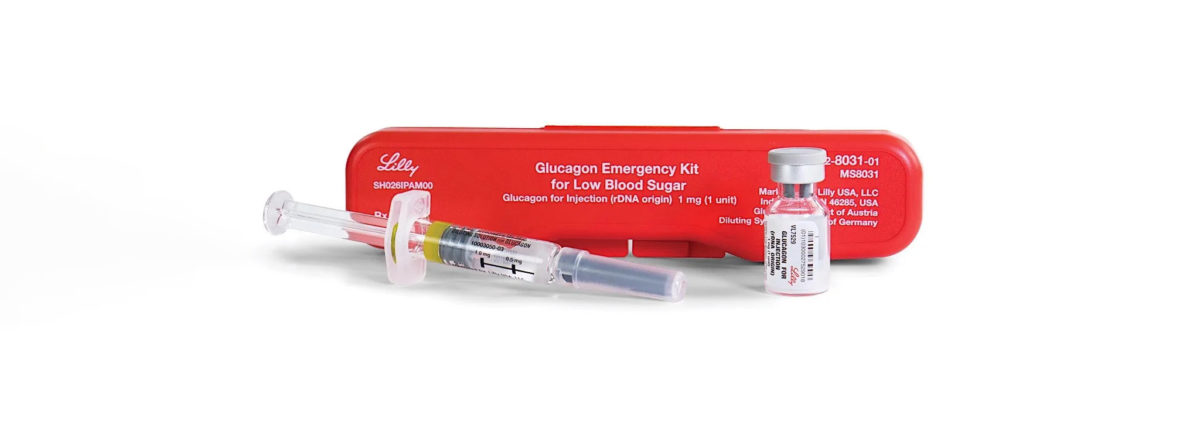
Lilly to Discontinue Old School Glucagon Emergency Kit at End of 2022
With several modern and ready-to-use glucagon options now available, Lilly will be discontinuing the old, red Glucagon Emergency Kit (GEK) in all markets. Please note: the generic GEK will still be available from Amphastar Pharmaceuticals—they have no plans to remove it from the market.
What this means for you:
If you take insulin and do not have (unexpired!) glucagon, it’s time to get one of the ready-to-use nasal spray or auto-injectable glucagons. These are the most modern forms of glucagon available today.
Glucagon is another hormone that people with diabetes cannot regulate properly. When ingested via nasal spray or injected in response to a severe low blood sugar, it quickly raises blood sugars to safe levels. It is a literal lifesaver.
Copay cards (for those with insurance) and patient assistance programs (for those without insurance) are available, most bringing your out-of-pocket cost down to $0 to $25.
What’s happening with the old red-case glucagon?
The phase-out of Lilly’s GEK will happen first in the U.S. on December 31, 2022.
In the U.S. market, several other glucagon options are now available including the nasal glucagon Baqsimi, the auto-inject devices Gvoke HypoPen and Zegalogue and the Gvoke Kit prefilled glucagon syringe.
The GEK was being prescribed in the U.S., Canada, Mexico, Argentina and Costa Rica. To ensure each market has access to a glucagon option, the GEK will be phased out in Mexico and Costa Rica after Lilly’s nasal glucagon Baqsimi becomes available.