Eversense from Senseonics: The First Implantable CGM
Written by: Beyond Type 1 Editorial Team
1 minute read
June 26, 2018
Could implantable technology be the future of continuous glucose monitoring? Meet the Eversense CGM, created by the device company Senseonics.
When the FDA approved the Eversense CGM by Senseonics, it marked the first time an implantable continuous glucose monitor (CGM) was cleared in the U.S. for adults aged 18 and older.
The Eversense has three key parts: a long-lasting sensor, a smart transmitter and a mobile app.
The standout feature is the sensor, which is implanted under the skin in the upper arm by a doctor.
Using advanced fluorescent tech, it measures blood sugar and sends the data to the transmitter, which sits on your skin. The transmitter then sends the info via Bluetooth to your phone.
While the sensor requires a doctor’s visit to replace every 90 days, a longer-lasting version—Eversense XL—is in the works, pending FDA approval.
For more information on the Eversense, review the PRECISE II study, which included 90 patients living with diabetes.
Why consider Eversense?
- 90-day continuous wear with no missed readings.
- Quick on-off transmitter without removing the sensor.
- Alerts via vibration, even if your phone isn’t nearby.
- Proven accuracy, rivaling or beating other CGMs.
- Requires quarterly doctor visits and added costs.
Read more about Continuous Glucose Monitors here.

Author
Beyond Type 1 Editorial Team
Beyond Type 1 is the largest diabetes org online, funding advocacy, education and cure research. Find industry news, inspirational stories and practical help. Join the 1M+ strong community and discover what it means to #LiveBeyond a diabetes diagnosis.
Related Resources

On March 4, Tandem Diabetes Care, maker of the Mobi insulin pump, announced an expanded...
Read more

As the years go on, CGMs (continuous glucose monitors) are becoming more and more visible....
Read more
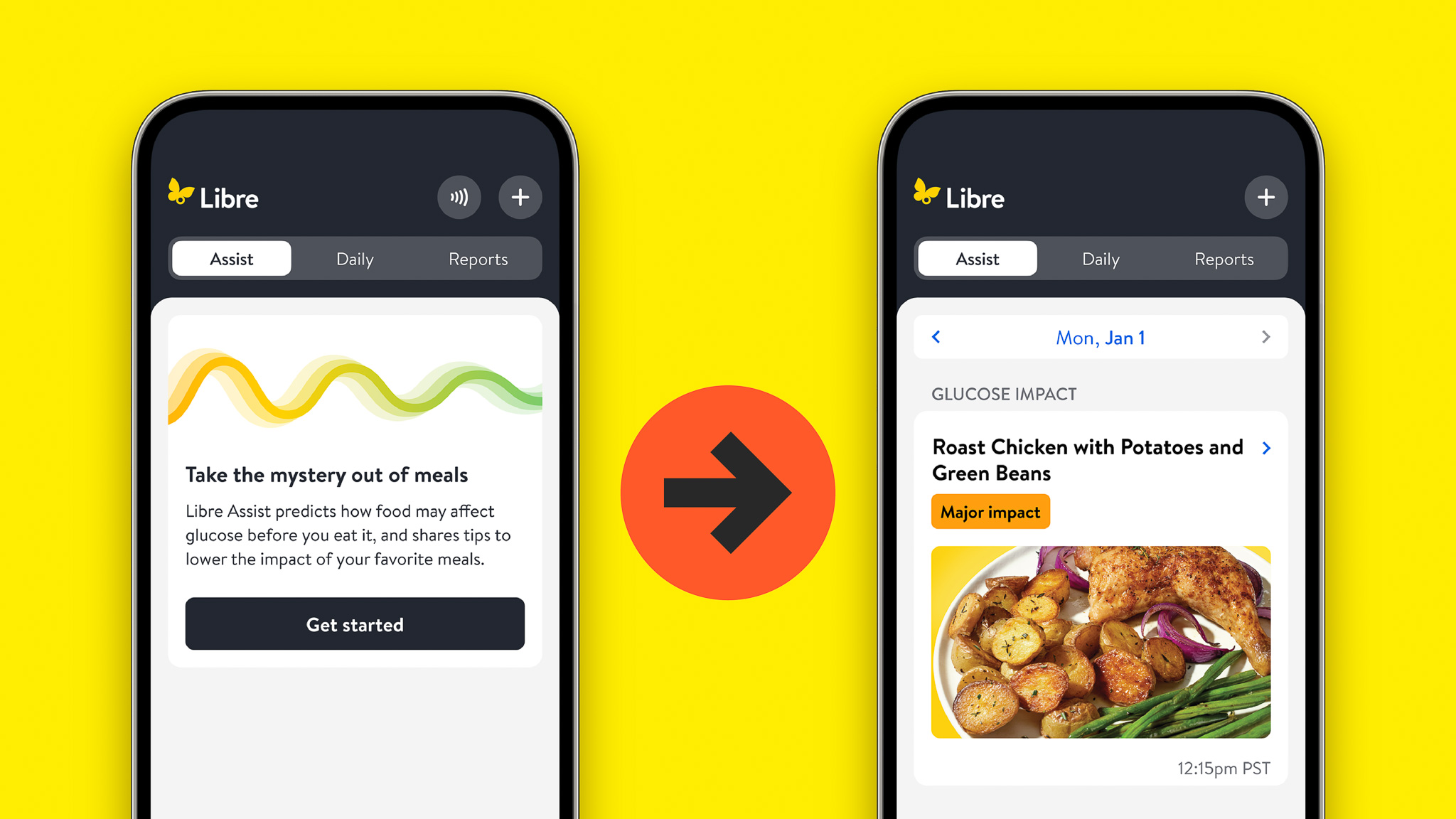
On January 5, Abbott released a new feature within their Libre continuous glucose monitoring (CGM)...
Read more

